Do we need antioxidant supplements?
Quick answer is no, provided that you eat natural food and you are physically and mentally active. What we really need to be joyfully healthy is as simple as this:
- – Diversity of natural and minimally processed food
- – Pure water
- – Oxygen diluted with nitrogen (air)
- – Physical and intellectual work daily
Everything we need for health is generated and balanced based on the above conditions. We are NOT free to eat whatever “fun food” surrogates food industry sells to us. To be healthy and happy we must make right nutritional choices. If this seems too boring, go ahead have fun with fast food, drink Coca-Cola, Kool-Aid, and stay on a couch watching TV in airless room. Then, yes, you will need a lot of vitamins, antioxidants, drugs for lowering blood cholesterol, drugs for lowering blood pressure, drugs for diabetes, drugs for cancer, etc., which will not really help anyway. Soon you will need wheelchair and life support too. The two reasons why we have vitamin and food supplements market flourishing are these:
- – Modern pseudo-economy increasingly plants food surrogates on us, pollutes water and air we need clean to survive
- – Popular ignorant thinking that nutritional deficiency caused by food surrogates can be compensated by taking food supplements afterwards. This fallacy keeps commercial value of supplements up.
As you can see, the existing monetary economy robs us twice at least. First, it profits from creating a problem by hooking us up on cheap health-damaging food which “tastes good.” Second, it profits from offering us a “solution” in the form of health-damaging drugs and useless supplements. Why are we surprised that our health keeps going down a drain? This looks like a new form of economic slavery to me.
The real solution is in clear understanding of what food is, also in rejecting food surrogates and making physically active lifestyle a common norm.
Each year, countless hopeful Americans shell out billions of dollars on antioxidant supplements (nearly $2 billion, in fact, just on beta carotene and vitamins C and E), believing they will dramatically lower their risk of cancer, heart disease, and memory loss. Most experts agree, however, that taking antioxidants is not a shortcut to good health or the answer to staying young.
Elements of antioxidant theory: Balance is critical
First of all, antioxidant is not a substance. Being an antioxidant rather is a role a molecule plays in a given situation. Therefore showing antioxidant activity is chemical property of a molecule.
Molecules are stable (low energy) combinations of two or more atoms which cling together due to sharing paired electrons. However, higher energy reactive intermediates bearing unpaired electrons, called radicals, are often formed in the course of chemical reactions, especially when oxygen, light or higher temperatures are involved. As the reactive radical particles are formed they do not stay alone for too long. Instead they “raid” virtually any surrounding molecules in proximity in order to pair their electron. Often the electron comes along with an atom (like hydrogen atom H) or a group of atoms thus causing severe damage to the donor molecule.
 Note that this is radical chain process in which another radical is formed that often is less aggressive and hence less damaging. The chain proliferation is stopped when two radicals meet together to form a stable molecule.
Note that this is radical chain process in which another radical is formed that often is less aggressive and hence less damaging. The chain proliferation is stopped when two radicals meet together to form a stable molecule.
Obviously, because oxygen and sunlight are the key elements of life, active radical intermediates can play either vital or fatal roles depending on how well the damage control mechanisms are functioning in living cells.
Oxygen and antioxidants are partners in reduction-oxidation (redox) processes resulting in the oxygen to gain an electron supplied by the antioxidant (reducing agent). Many important biochemical processes involve redox reactions. Cellular respiration, for instance, is the oxidation of glucose (C6H12O6) to CO2 and the reduction of oxygen to water. The summary equation for cell respiration is:
C6H12O6 + 6 O2 → 6 CO2 + 6 H2O
The process of cell respiration also depends heavily on the reduction of NAD+ to NADH and the reverse reaction (the oxidation of NADH to NAD+). Photosynthesis and cellular respiration are complementary, but photosynthesis is not the reverse of the redox reaction in cell respiration:
6 CO2 + 6 H2O + light energy → C6H12O6 + 6 O2
Photosynthesis involves the reduction of carbon dioxide into sugars and the oxidation of water into molecular oxygen. The reverse reaction, respiration, oxidizes sugars to produce carbon dioxide and water.
Oxidative stress reflects an imbalance between the systemic manifestation of reactive oxygen species (ROS) and a biological system’s ability to readily detoxify the reactive intermediates or to repair the resulting damage.
In humans, oxidative stress is thought to be involved in the development of cancer and a variety of other diseases. However, in the same time, reactive oxygen species can be beneficial, as they are used by the immune system as a way to attack and kill pathogens. Short-term oxidative stress may also be important in prevention of aging by induction of a process named mitohormesis.
Chemically, oxidative stress is associated with increased production of oxidizing species or a significant decrease in the effectiveness of cellular antioxidant defenses, such as glutathione, a substance synthesized in the human body from amino acids taken with food.
The oxygen molecule is itself a di-radical by nature because it has two unpaired electrons. This leads to formation of free radical that is very common in biology, the superoxide radical anion charged negatively. It is formed by transfer of one electron from an electron donor (reducing agent) to an oxygen molecule. Reduction is the opposite of oxidation, and occurs when an atom or molecule accepts one or more electrons. So we have O2 (di-oxygen molecule) reducing by adding an electron with the electron’s negative charge giving the superoxide anion -.O2.
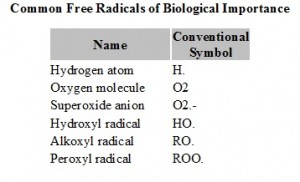 Another di-oxygen type molecule, hydrogen peroxide also can catch an electron and split at the HO-OH bond to produce hydroxyl radical HO. and hydroxyl anion (hydroxide) –OH. The hydroxyl radical, HO. is an extremely aggressive free radical. It reacts with almost everything as fast as it collides. So in other words HO. reacts almost every time it bumps into another molecule, generally to pull off a hydrogen atom and make water and also produce another free radical in the organic residue (See the above schemes). Hence such ROS may randomly damage DNA, enzymes, lipids, etc., and any other biomolecules thus triggering cancer and a variety of other health damaging processes in the human body.
Another di-oxygen type molecule, hydrogen peroxide also can catch an electron and split at the HO-OH bond to produce hydroxyl radical HO. and hydroxyl anion (hydroxide) –OH. The hydroxyl radical, HO. is an extremely aggressive free radical. It reacts with almost everything as fast as it collides. So in other words HO. reacts almost every time it bumps into another molecule, generally to pull off a hydrogen atom and make water and also produce another free radical in the organic residue (See the above schemes). Hence such ROS may randomly damage DNA, enzymes, lipids, etc., and any other biomolecules thus triggering cancer and a variety of other health damaging processes in the human body.
Your natural damage control
The best studied cellular antioxidants to fight free radicals are the cellular enzymes superoxide dismutase (SOD), catalase, and glutathione peroxidase. Once oxidized, glutathione can be reduced back by glutathione reductase, using NADPH as an electron donor.
From the above, three remarkable consequences follow:
- – Your most powerful natural antioxidant, glutathione is not an essential nutrient. It is synthesized in your body from scratch: the amino acids L-cysteine, L-glutamic acid, and glycine
- – Once glutathione has accomplished its mission on neutralizing active terrorist radicals, it is not wasted! Instead, glutathione is restored back to its full antioxidant capability
- – All it takes to keep your body protected from the oxidative damage is to eat normal human food and exercise
The ratio of reduced glutathione to oxidized glutathione within cells is often used as a measure of cellular toxicity. Less well studied (but perhaps just as important) enzymatic antioxidants are the peroxiredoxins and the recently discovered sulfiredoxin. Other enzymes that have antioxidant properties (though this is not their primary role) include paraoxonase, glutathione-S transferases, and aldehyde dehydrogenases.
Please note that these natural cellular antioxidants act as complexes with proteins and thus are quite different from individual phenolic or flavonoid antioxidant substances isolated from natural sources.
Antioxidants: supplements and potential hazards
Oxidative stress (as formulated in Harman‘s free radical theory of aging) is also thought to contribute to the aging process. However recent evidence from Michael Ristow‘s laboratory suggests that oxidative stress may also promote life expectancy of Caenorhabditis elegans by inducing a secondary response to initially increased levels of reactive oxygen species.
According to the article Antioxidants: Beyond the Hype published recently by Harvard school of public health: “Antioxidants came to public attention in the 1990s, when scientists began to understand that free radical damage was involved in the early stages of artery-clogging atherosclerosis and may contribute to cancer, vision loss, and a host of other chronic conditions. Clinical trials began testing the impact of single substances, especially beta-carotene and vitamin E, as weapons against heart disease, cancer, and the like.
Often the claims have stretched and distorted the data: While it’s true that the package of antioxidants, minerals, fiber, and other substances found naturally in fruits, vegetables, and whole grains help prevent a variety of chronic diseases, it is unlikely that high doses of antioxidants can accomplish the same feat.”
Recent meta-analysis conducted in 2007 suggested that popular antioxidant supplements such as Vitamin A, Beta Carotene, and Vitamin E may increase mortality risk in low-risk group humans (although they decreased risk in the high-risk group).
As it was pointed out above, glutathione is an abundant natural tripeptide found within almost all cells. Glutathione is highly reactive and is often found conjugated to other molecules. It plays several vital roles within a cell including antioxidation, maintenance of the redox balance, modulation of the immune response, and detoxification of xenobiotics. However, with respect to cancer, glutathione metabolism is able to play both protective and pathogenic roles. By conferring resistance to a number of chemotherapeutic drugs, elevated levels of glutathione are able to protect tumor cells in bone marrow, breast, colon, larynx and lung cancers. Studies investigating the role of glutathione in promoting cancer, impeding chemotherapy, and the use of glutathione modulation to enhance anti-neoplastic therapy were presented recently (Cell Biochem Funct. 2004 Nov-Dec;22(6):343-52. Duarte, CA).
Even more adverse effects should be expected from the consumption of synthetic phenolic antioxidant preservatives such as BPA, BHT and TBHQ broadly used in food industry. For instance, TBHQ is a highly effective antioxidant. The FDA sets its upper limit to 0.02% of the oil or fat content in foods. At higher doses, it has some negative health effects on lab animals, such as producing precursors to stomach tumors and damage to DNA. A number of studies have shown that prolonged exposure to high doses of TBHQ may produce carcinogenic effect. This is the point where the controversy, discrepancy and confusion come from:
The oxidative characteristics and/or metabolites of ANTIOXIDANTS may contribute to carcinogenicity or tumorigenicity; however the same factors may also combat oxidative stress.
If antioxidants were harmless, it wouldn’t much matter if you took them “just in case.” A few studies, though, have raised the possibility that taking antioxidant supplements, either single agents or combinations, could interfere with health. The first inkling came in a large trial of beta-carotene conducted among men in Finland who were heavy smokers, and therefore at high risk for developing lung cancer.
Conclusions
The conclusions are not new.
- – Think of this absurd of our reality: In the economy where abstract numbers of profit and GDP are valued higher than actual parameters of people’s health and well-being, no one even dare to set a goal of achievement of healthy life for all and everyone. Healthy society would mean elimination of pharmaceutical and health insurance industries, and many questionable health services altogether. This would mean an unsustainable blow to the modern “economy” which promotes consumerism and sickness. Therefore we cannot expect any serious improvements in public health within the present ill-conceived economic paradigm
- – Deceptive abundance of cheap food brought about for the sake of profits by its mass-production has come at a price of massive chronic malnutrition and undermined overall health of generations of consumers. Millions of suffering consumers, in their desperation aggravated by lack of education, often become victims of yet another fallacy – the overblown benefits of the means of affordable alternative medicine; sometimes even clear loss of common sense is not an obstacle. For instance, the concept of usefulness of antioxidant supplements for prevention or fighting cancers co-exists with the irresponsible beliefs of benefits of drinking hydrogen peroxide oxidant (the hydroxyl radical generator!) for cancer treatment.
Our bodies have remarkably efficient “built-in” cyclic mechanisms of redox balance and oxidative damage control. All we need to do is let them work properly without being compromised by excessive consumption of sugars and lacking of physical activity or insanity of drinking hydrogen peroxide. Abundant food and sugars are good only in hands and stomachs of EDUCATED consumers who understand that every bite of it must be “earned” by your physical and intellectual work. Eating whole fruits, vegetables, and whole grains – all rich in varieties of antioxidants and their enzyme complexes – provides protection against many diseases and scourges of aging.
Category: Food and cancer, Healthy diet, Supplements









I’m gone to say to my little brother, that he should also go to see this
weblog on regular basis to take updated from newest reports.